 Click here for a full size version of this image.
|
During magnetic storms, the glow may move southwards, and on occasion it can be seen in much of the US. It often appears as a glow on the horizon, like the glow preceding sunrise, and has therefore become known among scientists as "aurora borealis" ("aurora" for short), Latin for "northern dawn." A similar phenomenon is also seen in southern polar regions.
|


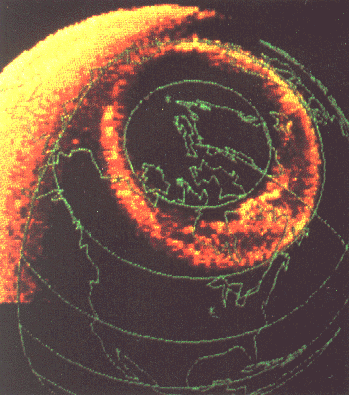
 Red Aurora
Red Aurora
 Official GSFC Home Page
Official GSFC Home Page  NASA WWW Home Page
NASA WWW Home Page